Dalit Sela-Donenfeld

Developmental Biology, Molecular Embryology, Neural Development, Embryotoxicity
 Special interest
Special interest
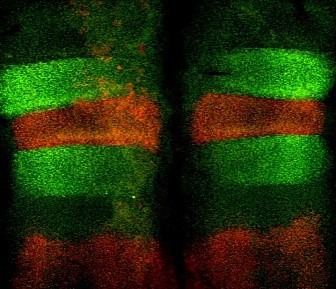
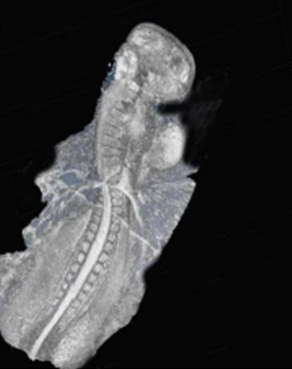
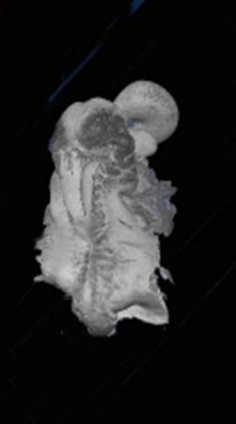
Developmental Biology; mechanisms underlying hindbrain development, neural progenitor/stem cells, differentiation and assembly of neural circuits; effectors that regulate neural crest cell migration; Extracellular Matrix, Matrix metalloproteases, Environmental pollutants and embryotoxicity.
|

|
|
|